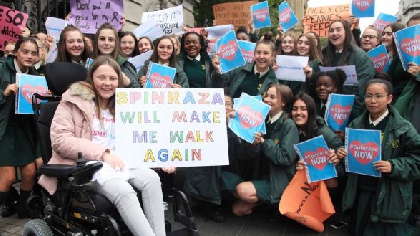
The Minister for Health, Wicklow TD, Simon Harris says he has been assured that there will be no delay in accessing the drug Spinraza, following a lengthy campaign by parents of those affected.
The HSE’s leadership team has approved access to the drug for children with type 1, 2 and 3 Spinal Muscular Atrophy on an exceptional and individualised basis.
Earlier this year, the HSE said the drug would cost about €600,000 in the first year to treat each of the 25 Irish children with the disease and €380,000 a year thereafter,.
Spinraza is now approved in 26 countries around Europe with just Estonia rejecting it.


 Tributes Paid To Former Wicklow GAA Manager Mick O' Dwyer Following His Death Aged 88
Tributes Paid To Former Wicklow GAA Manager Mick O' Dwyer Following His Death Aged 88
 Planning Application Lodged To Turn Wicklow Pub Into IPAS Centre
Planning Application Lodged To Turn Wicklow Pub Into IPAS Centre
 Inquest Hears 2 Year Old Girl Died Of Brain Damage Following Choking Incident At Greystones Creche
Inquest Hears 2 Year Old Girl Died Of Brain Damage Following Choking Incident At Greystones Creche
 Closure of Herbert Road Junction In Bray Confirmed To Facilitate Bus Corridor
Closure of Herbert Road Junction In Bray Confirmed To Facilitate Bus Corridor
 Annual Spring Clean Month Arrives Across County Wicklow
Annual Spring Clean Month Arrives Across County Wicklow




